
The Science of Delta-Nine.
Cannabis is pharmacologically complex — powerful, but historically unpredictable.
Delta-Nine was built to change that.
We do not rely on strain folklore or broad categories.
We constructed a structured control system grounded in thermodynamics, pharmacokinetics, receptor pharmacology, and neurophysiology — designed to translate plant chemistry into reproducible outcomes.
To achieve predictability in medical and therapeutic cannabis use, five domains must be controlled:
01. Compound Release Control
Natural flower releases compounds across a temperature spectrum.
Structured sequencing shapes release over time.
Establishes temporal control.
-
Different compound classes volatilise at distinct temperature ranges, altering the order in which cannabinoids and terpenes enter systemic circulation.
Sequencing temperature therefore changes onset velocity, receptor exposure timing, and experiential trajectory.
Within Delta-Nine, this principle is structured as ThermoTemporal Vaporisation (TTV™) — a defined phase model for controlled release architecture.
02. Delivery Physics
Hardware alters compound transfer.
Heat curves, airflow, and packing geometry affect dose behaviour.
Establishes physical control.
-
Thermal inertia, airflow dynamics, and chamber geometry determine how evenly heat is distributed and how efficiently vapour is extracted.
These variables alter volatilisation rate, transfer efficiency, and effective dose.
Within Delta-Nine, Delivery Profiles formalise device behaviour to preserve model fidelity.
03. Predictive Psychoactive Modelling
Psychoactivity evolves dynamically.
Absorbed load and temporal progression can be modelled.
Establishes predictive control.
-
Description text goes here
04. Compositional Control
Natural flower releases compounds across a temperature spectrum. Uncontrolled heating produces overlapping, inconsistent effects.
-
Cannabinoid and terpene ratios determine receptor weighting and experiential bias.
By calculating milligram content per gram and adjusting blend weights accordingly, composite chemotypes can be constructed with defined proportions.
Within Delta-Nine, this structured approach is termed Profile Blending.
05. Neurophysiological State Regulation
Natural flower releases compounds across a temperature spectrum. Uncontrolled heating produces overlapping, inconsistent effects.
-
Cannabinoids modulate CB1 receptors embedded within active neural networks; their effects depend on pre-existing cortical and autonomic state.
Arousal level, expectation pathways, and network activity influence how psychoactivity is amplified, attenuated, or integrated.
Within Delta-Nine, structured behavioural and contextual alignment is formalised as BioModulations™.

Delta-Nine is a constructed, internally coherent system.Grounded in pharmacology, thermodynamics, molecular kinetics, and behavioural psychology, it organises cannabis dry-herb vaporisation into a structured, reproducible method of targeted pharmacological modulation.The Case for a Cannabis Framework.
Unique pharmacology demands bespoke structure.Cannabis is not a single pharmacological agent.
It is a multi-compound system with distributed receptor activity.
More than one hundred cannabinoids and terpenes interact across:
• CB1 receptors within the central nervous system
• CB2 receptors in immune and peripheral tissues
• TRP channels involved in nociception and inflammation
• Serotonergic and GABAergic pathways influencing mood and cognition
Unlike conventional medicines, inhaled cannabis allows real-time user modulation.
Dose magnitude, temperature selection, and inhalation pattern directly shape absorption kinetics and psychoactive trajectory.
This combination — multi-pathway activity plus user-adjustable delivery — makes cannabis uniquely powerful, and uniquely variable.
The problem is not the plant.
It is the absence of structured delivery.
That is what Delta-Nine provides.
01. Psychoactivity is a primary mechanism — not a side-effectCannabis does not merely relieve symptoms; it alters perception, cognition, mood, and attention through CB1 receptor activation in the central nervous system. These psychoactive effects are intrinsic to its therapeutic action.
-
CB1 receptor density in cortical and limbic regions explains why THC influences executive function, emotional processing, and memory encoding.
Therapeutic outcomes are inseparable from these psychoactive shifts.
02. Multi-pathway activity requires precisionCannabis does not act on a single receptor or pathway.
Its compounds engage central and peripheral systems simultaneously, producing layered and sometimes competing effects.
Small shifts in compound ratios or dose magnitude can alter downstream signalling patterns, changing both therapeutic outcome and subjective experience.
Precision is therefore not optional — it is foundational.
-
Cannabinoids exhibit partial agonism at CB1 and CB2 receptors, while terpenes and minor cannabinoids influence ion channels and neurotransmitter systems. These interactions generate non-linear dose–response relationships that require structured modelling.
03. Thermal release alters pharmacological profileCannabinoids and terpenes transition into vapour across temperature ranges rather than fixed boiling points. Within the resin matrix, intermolecular forces broaden and overlap these ranges.
Altering temperature does not merely increase intensity.
It changes which compounds are delivered, and in what sequence.
Structured thermal progression transforms this variability into defined pharmacological phases.
-
Selective heating influences decarboxylation kinetics, terpene preservation, and cannabinoid volatilisation ratios. Sequential temperature control enables temporal shaping of psychoactive trajectory.
04. User-controlled dosing demands modellingInhaled cannabis allows immediate titration.
Users directly influence dose magnitude, delivery timing, and session progression.
Rapid pulmonary absorption produces fast plasma THC elevation, followed by dynamic redistribution and metabolic decline.
Without modelling, variability dominates.
With modelling, intensity, duration, and direction become predictable.
-
Peak plasma THC typically occurs within minutes of inhalation. The psychoactive curve that follows reflects absorbed dose, distribution kinetics, and receptor modulation — making time-domain analysis essential.

Cannabis is commonly categorised as a recreational drug.
Pharmacologically, that definition is insufficient.
It functions as a user-adjustable neuromodulatory system.
Reframing Cannabis
Through controlled dose, compound selection, and thermal sequencing, cannabis enables modulation of mood, perception, nociception, appetite regulation, and sleep architecture — in real time.
No other medicine permits this degree of real-time, user-directed nervous-system modulation.
The distinction is not recreational versus medical.
It is structured versus unstructured use.
ThermoTemporal Vaporisation (TTV™)
ThermoTemporal Vaporisation (TTV™) is Delta-Nine’s structured method for controlling cannabis compound release across temperature and time.
It is not a device.
It is not a preset.
It is a thermodynamic framework.
TTV™ recognises that cannabinoids and terpenes do not release as a single event.
They transition in overlapping bands influenced by temperature, matrix interactions, airflow, and sequence.
By shaping heat progression deliberately, TTV™ converts chemical variability into defined pharmacological phases.
Temperature becomes timing.
Timing becomes structure.
Structure becomes predictable modulation.
Conventional guidance reduces vaporisation to a binary:
Low temperature for flavour / High temperature for potency.
This is directionally correct — but chemically incomplete.
Cannabis resin contains dozens of active compounds with distinct volatility curves and receptor profiles.
Unstructured heating collapses them into noise.
We developed TTV™ to separate signal from noise — to transform a multi-compound plant into a controllable, phase-based delivery system.
TTV™ is the bridge between plant chemistry and reproducible outcome.

TTV: Beyond Flavour-Potency Balance.
Chemical Basis of ThermoTemporal Vaporisation (TTV™)
01.
Cannabis resin is not a single active compound.
It is a thermally responsive mixture of cannabinoids, terpenes, and minor constituents embedded within a lipid-rich matrix.
Each compound exhibits:
A characteristic vapour pressure curve
A temperature-dependent volatilisation range
Distinct receptor binding behaviour
Unique pharmacodynamic contribution
In isolated laboratory conditions, boiling points are sharp.
Within plant resin, intermolecular interactions broaden these into overlapping release bands.
As temperature increases, compound dominance does not rise uniformly.Relative concentration ratios shift as lighter fractions deplete and heavier fractions emerge.The psychoactive trajectory therefore evolves as a function of sequence, not simply peak temperature.

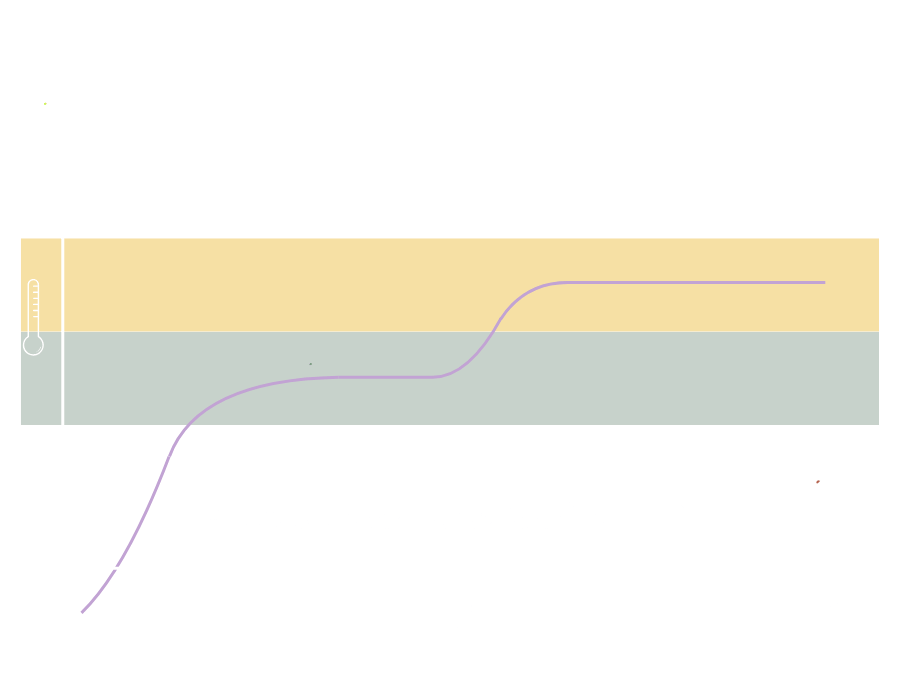
Key Principles of TTV™-
At defined temperature intervals, clusters of compounds become proportionally dominant in the vapour stream.
The psychoactive profile changes not only in magnitude, but in qualitative direction.
-
Increasing temperature alters the relative ratios of cannabinoids and terpenes delivered per inhalation.
Effect modulation results from ratio change, not simply dose increase.
-
When thermal progression is organised into discrete phases, pharmacological emphasis can be shifted within a single session.
Cognitive activation, somatic modulation, and sedative loading can be sequenced deliberately.
-
Unstructured heating compresses release into a narrow time window.
Structured sequencing distributes compound delivery over time, reducing abrupt psychoactive spikes.
-
Without structure, vaporisation produces variable compound mixtures and unpredictable psychoactive trajectories.
With structure, temperature becomes:
A reproducible control variable governing delivered chemistry.
TTV™ transforms vaporisation from extraction into modulation.

02.
Pharmacodynamic Basis of Phase Modulation
Cannabis effects do not arise from THC alone.
They emerge from multi-receptor interaction, dose-dependent binding dynamics, and compound ratio shifts across time.
As TTV™ alters vapour composition phase by phase, it changes not only intensity — but receptor weighting.
Thermal sequencing therefore becomes pharmacodynamic sequencing.
Receptor-Level Mechanisms-
Δ9-THC acts as a partial agonist at CB1 receptors.
Partial agonism produces non-linear psychoactive scaling:
effect does not increase proportionally with dose.
As compound ratios shift across phases, CB1 activation strength and distribution change — influencing cognition, perception, and affective tone.
-
Cannabinoids interacting at CB2 contribute to anti-inflammatory and somatic modulation.
Phase-dependent shifts in cannabinoid profile alter peripheral signalling without necessarily increasing central psychoactivity.
-
Certain terpenes and cannabinoids activate transient receptor potential (TRP) channels involved in nociception, thermoregulation, and inflammatory signalling.
As lighter terpene fractions deplete and heavier fractions emerge, TRP engagement evolves across the session.
-
Cannabinoid signalling indirectly modulates serotonin and GABA transmission.
This contributes to:
• Anxiolytic or anxiogenic shift
• Sedative loading
• Cognitive quieting
• Mood elevation
These changes are ratio-dependent, not purely dose-dependent.
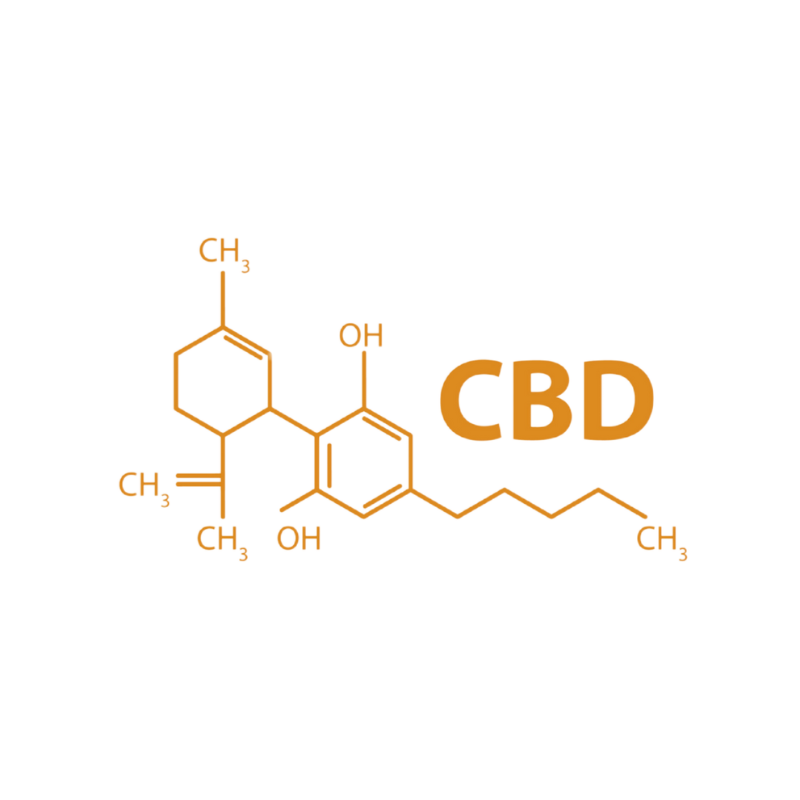
CBD as a Modulatory Layer
CBD does not simply counteract THC.
It functions as a negative allosteric modulator at CB1 receptors and influences multiple non-cannabinoid pathways, including serotonin (5-HT1A) and TRP channels.
When present within the vapour stream:
• CB1 signalling amplitude is attenuated
• Psychoactive sharpness may soften
• Temporal curves may smooth
• Anxiogenic peaks may reduce
CBD therefore alters the shape and direction of the psychoactive trajectory — not merely its intensity.
In structured sequencing, CBD becomes a key tuning variable within the delivery architecture.
-
If compound ratios determine receptor weighting,
and temperature determines compound ratios,
then structured temperature progression determines pharmacodynamic direction.
TTV™ converts thermochemical shifts into controlled receptor-level modulation.
Effect becomes architecture, not accident.
∆9E™ Psychoactivity Modelling
The first algorithmic model to predict how cannabis feels in real time.03.

TTV™ provides control of delivered chemistry.
Δ9E™ translates that chemistry into predicted pharmacodynamic behaviour over time.
The central question is not simply:
What compounds are present?
It is:
How do they interact within the body, and how does that interaction evolve across minutes and hours?
Δ9E™ converts chemical inputs into structured psychoactivity curves.
What Δ9E™ Produces
The output is not a scalar value.
It is a trajectory. A curve representing:
• Intensity (magnitude over time)
• Vector Direction (Δ9V — temporal effect shift)
• Duration (temporal persistence)
• Total Psychoactive Load (AUC; ψ-minutes)
Psychoactivity becomes modelled, graphable, comparable.
And reproducible.
Δ9E™ integrates five domains of data:
• ΔPot-Q — effective cannabinoid load (potency × quantity × bioavailability)
• Terpene Modulation — ratio-dependent neuromodulatory weighting
• ΔTVP-Mod — thermodynamic phase sequencing and release distribution
• ΔOnset-T — temporal absorption and rise kinetics
• Δ9Rx Clinical Mapping — alignment of intensity bands with therapeutic targets
These variables are mathematically weighted and resolved into a time-based psychoactivity curve.
Worked Example
Type IA flos. (20% THC, <1% CBD)
With and Without CBD ModulationWe compare:
Unmodulated Type IA
Type IA under added CBD modulation (via terpene-aligned Type III flower)
20% ∆9-THC (from THCA); <1% CBD Limonene – Pinene – Myrcene dominant

0.1% ∆9-THC (from THCA); 12% CBD Limonene – Pinene – Myrcene dominant

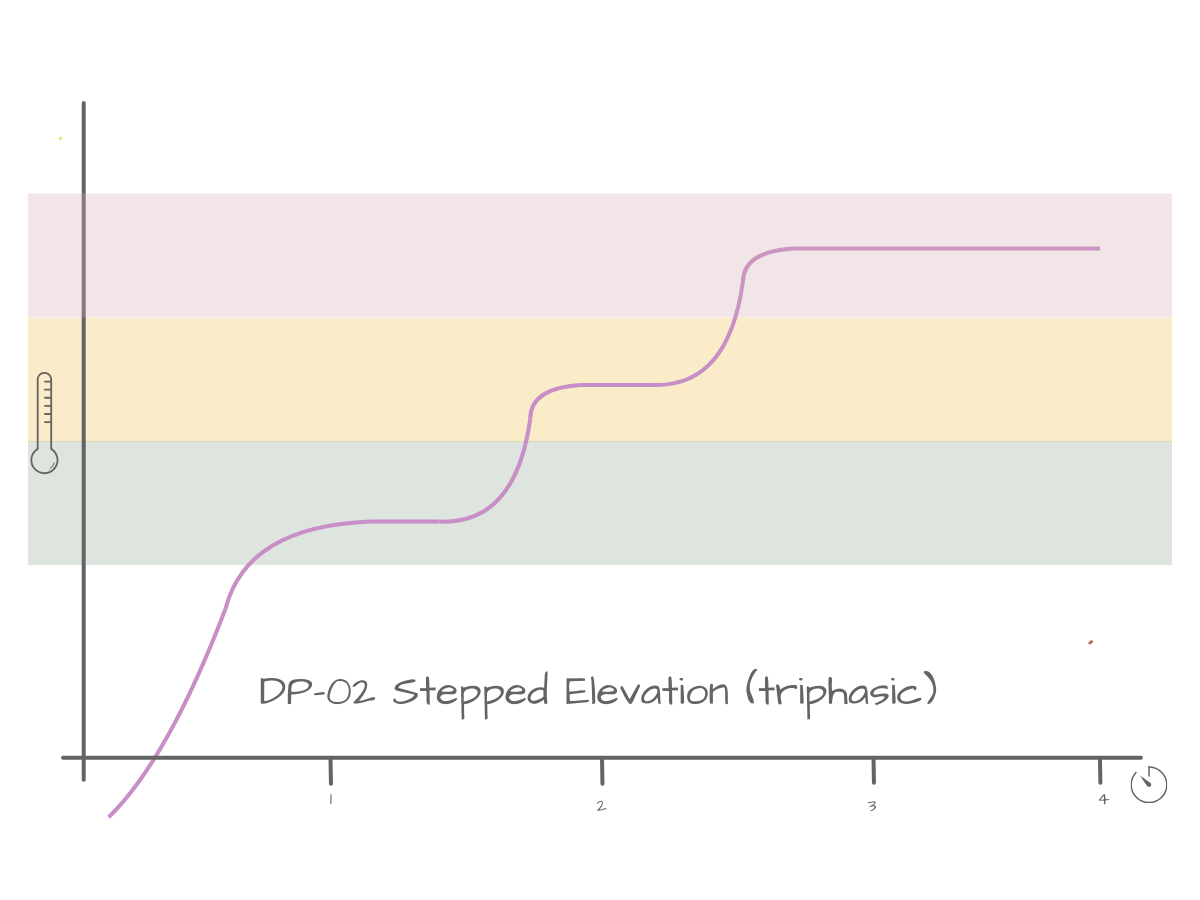
Delivery Profile:
DP-02 Stepped Elevation (Triphasic)
Structured over ~4 minutes.-
NeuroPriming™ Thermal Band
Chamber temperature rises into the lower defined band and stabilises.
Within this range, vapour composition is biased toward:
• Limonene
• α-Pinene
• Early THC fraction
This is a thermodynamic plateau — a controlled temperature hold: It does not represent effect intensity. However, the chemistry released during this hold begins to accumulate systemically:
• Early CB1 engagement
• TRP channel activation
• Cortical bias toward alertness
Plasma THC is rising. Nothing resets when Phase II begins.
-
NeuroLift™ Thermal Band
The delivery profile steps upward into a higher thermal band and stabilises.
This controlled temperature increase shifts vapour composition toward:
• Increased THC fraction
• Residual limonene/pinene
• Emerging myrcene
Again, the plateau represents thermal stability, not experiential flatness.
Pharmacodynamically (modelled in Δ9E™), this phase typically corresponds to:
• Increasing CB1 occupancy
• Continued cortical activation
• Early somatic weighting
The chemistry from Phase I remains active. Phase II adds to it.
Stacking, not replacement.
-
Somatic Optimisation™ Thermal Band
A second controlled temperature increase moves the chamber into the higher band.
Temperature stabilises again.
Vapour composition now favours:
• Higher THC fraction
• Myrcene dominance
• Reduced lighter terpene presence
This is the final thermal plateau of the delivery profile.
The psychoactive consequences of this shift — increased CB1 amplitude, greater GABAergic weighting, deeper somatic bias — are not shown in the DP graph.
They are predicted in the Δ9E™ curve.
The psychoactive trajectory — intensity, direction, duration — is resolved in Δ9E™ by integrating:
• ΔPot-Q (effective cannabinoid load)
• ΔTVP-Mod (phase-sequenced release weighting)
• ΔOnset-T (absorption kinetics)
• Terpene modulation
• CBD modulation (if present)
Interpreting Ψt Curves

The curves above represent modelled psychoactive intensity (ψ) over time under identical delivery conditions.
The thermal input (DP-02) is unchanged.
The divergence arises from altered cannabinoid ratio and receptor-level modulation.
CBD does not eliminate psychoactivity.
It reshapes trajectory — compressing amplitude and reducing unpredictability.
AUC - Total Psychoactive Loading
Beyond peak intensity, Δ9E™ resolves total psychoactive exposure as Area Under the Curve (AUC), expressed in ψ-minutes.
AUC is calculated as the absolute integral of ψ(t):
Total psychoactive load = ∫ |ψ(t)| dt
This metric captures cumulative effect exposure across time, independent of whether ψ is positive (stimulating bias) or negative (somatic bias).


In this example:• Type IA exhibits greater total AUC, reflecting higher overall psychoactive load.• The reformulated Type II exhibits reduced AUC, with load redistributed across a smoother trajectory.Peak intensity and total load are not interchangeable. A lower peak can still produce meaningful exposure if distributed across time.A higher peak does not necessarily imply stability.-
• Rapid ascent to higher peak ψ
• Greater early-phase volatility
• Deeper post-peak trough
• Larger amplitude oscillation
-
• Moderated peak ψ
• Reduced overshoot
• Shallower trough
• Smoother recovery profile

04.
Compositional Control I:
Profile Blending for Cannabinoids
Single cannabis flowers express fixed cannabinoid and terpene ratios.
These ratios determine receptor weighting, modulation pathways, and experiential bias. In practice, most available flowers cluster around high-THC dominance or limited balanced ratios.
Compositional control expands beyond these fixed chemotypes.
+
Profile Blending is the structured construction of target cannabinoid and terpene proportions using calculated milligram inputs.
Rather than selecting strains by name, outcomes are built through defined chemical proportions — enabling deliberate shaping of magnitude, bias, and balance.
The Arithmetic of Construction
Cannabinoid percentages convert directly into milligram content via a simple formula:
For example:
0.15 g of a 24% THC flower contains:
150 mg total material × 0.24 = 36 mg THC
By adjusting the relative weights of two or more flowers, defined ratios can be constructed.
Milligram Conversion Rule:Cannabinoid (mg) = mass (g) × Concentration (%) × 10
Qualitative Shift
The example below shows how the properties of two different chemovars can be deliberately combined for moderated effect:
Type I flos.
High THC, low/no CBD
0.10 g of a 24% THC flower
= 0.1 x 24 x 10 = 24mg THC
0.10 g of 8% THC / 8% CBD flower
Type II flos.
Balanced THC:CBD
= 0.1 x 8 x 10 = 8mg THC
+ 0.1 x 8 x 10 = 8mg CBD
↪ Moderated psychoactivity, increased somatic tone, reduced intensity spike.
Composite flos.
Custom cannabinoid balance
͢
= 32 mg THC + 8 mg CBD in 0.2g flower
Converting back to %:
16.5% THC + 4% CBD; or
0.2g of a 4:1 composite at T16.5 : C4
↪ Preserved psychoactive clarity with moderated peak intensity and reduced volatility.
Target Ratio Construction
(Exact Method)
Flower 1:
THC = a%
CBD = b%
Flower 2:
THC = x%
CBD = y%
Target ratio:
Tt : Ct
Total blend mass:
m grams
-
mg per gram = % × 10
So:
Flower 1
THC₁ = 10a
CBD₁ = 10b
Flower 2
THC₂ = 10x
CBD₂ = 10y
-
Then (m − w) = grams of Flower 2
Total THC in blend:
THC_total = w(10a) + (m − w)(10x)
Total CBD in blend:
CBD_total = w(10b) + (m − w)(10y)
-
We require:
THC_total / CBD_total = Tt / Ct
So:
[w(10a) + (m − w)(10x)]
——————————————— = Tt / Ct
[w(10b) + (m − w)(10y)]
The factor of 10 cancels, so simplify:
[w a + (m − w) x]
——————————————— = Tt / Ct
[w b + (m − w) y]
-
After rearranging:
w = m × (Tt·y − Ct·x)
———————————————
(Tt·y − Ct·x) + (Ct·a − Tt·b)
That is the closed-form solution.
Once w is known:
Flower 1 weight = w
Flower 2 weight = m − w
-
Flower 1: 24% THC ; 0% CBD
Flower 2: 8% THC ; 8% CBD
Target ratio: 4 : 1
Total blend: m = 0.20 g
Plug into formula:
w = 0.20 × (4·8 − 1·8)
———————————————
(4·8 − 1·8) + (1·24 − 4·0)
= 0.20 × (32 − 8)
———————————————
(32 − 8) + 24
= 0.20 × 24 / 48
= 0.10 g
So:
0.10 g Flower 1
0.10 g Flower 2
As previously demonstrated.
If Flower 1 is high-THC and Flower 2 contains CBD
Then:
– Increasing Flower 2 proportion lowers the ratio toward 1:1.
– Increasing Flower 1 proportion raises the ratio toward THC dominance.
For most practical blends, iterative adjustment within 5–10% increments achieves the desired ratio rapidly.
Commercial & medical cultivars cluster at high-THC dominance or near-balanced ratios:
Flos. type
Typical THC
Typical CBD
Common Ratio
III
≤1%
10–20%
1:10+
↪ Rapid onset, strong CB1 activation, higher anxiety potential at dose.
20:1+
I
18-30%
≤1%
1:1 – 2:1
II
5–12%
5–12%
Ratios such as 3:1 · 4:1 · 5:1 · 7:1 rarely exist as standalone chemovars and will require formulation as per the above.
05.Neurophysiological Modulation
Cannabis alters brainwave activity, network dynamics, and autonomic tone.
Breathing, sound, aromatics, and attentional practices act on overlapping pathways.
Coordinated inputs shape state trajectory and experiential integration.
Establishes organism-level modulation.

Cannabinoid receptor modulation intersects with autonomic regulation, cortical oscillation, and limbic circuitry. These same pathways are accessible through non-chemical inputs.
Neurophysiological state is not fixed.
Multiple non-chemical inputs converge on the same pathways influenced by cannabinoids.
Structured coordination transforms isolated effects into engineered state transitions.
Cross-Modal Pathway Convergence
Breathing patterns, posture, and thermal input alter vagal tone and sympathetic–parasympathetic balance.
Autonomic Regulation

Limbic Activation

Olfactory pathways access amygdala and hippocampal circuits directly, modulating emotional weighting.
Auditory rhythm and pulsed stimuli bias cortical oscillatory synchronisation and temporal coherence
Rhythmic Entrainment
Attentional practices recalibrate Default Mode and salience network dynamics, shaping interpretation and integration.
Cognitive Structuring

Light frequency, stroboscopic stimulation, and gaze anchoring influence cortical excitation patterns and circadian signalling.
Visual-Cortical Input

Emerging electrical stimulation methods alter cortical excitability thresholds and network responsiveness.
Direct Neuromodulation

Cannabis does not act in isolation.
Psychoactive outcome emerges from compound delivery interacting with dynamically modifiable neural systems.
State is not background.
It is an active variable.
04. Compositional Control II:
Profile Blending for Terpenes
Proportional Volatile Engineering
Terpene composition determines directional character.
Weighted mass blending produces a new volatile architecture, flattening extremes and reshaping experiential bias without altering overall terpene percentage.
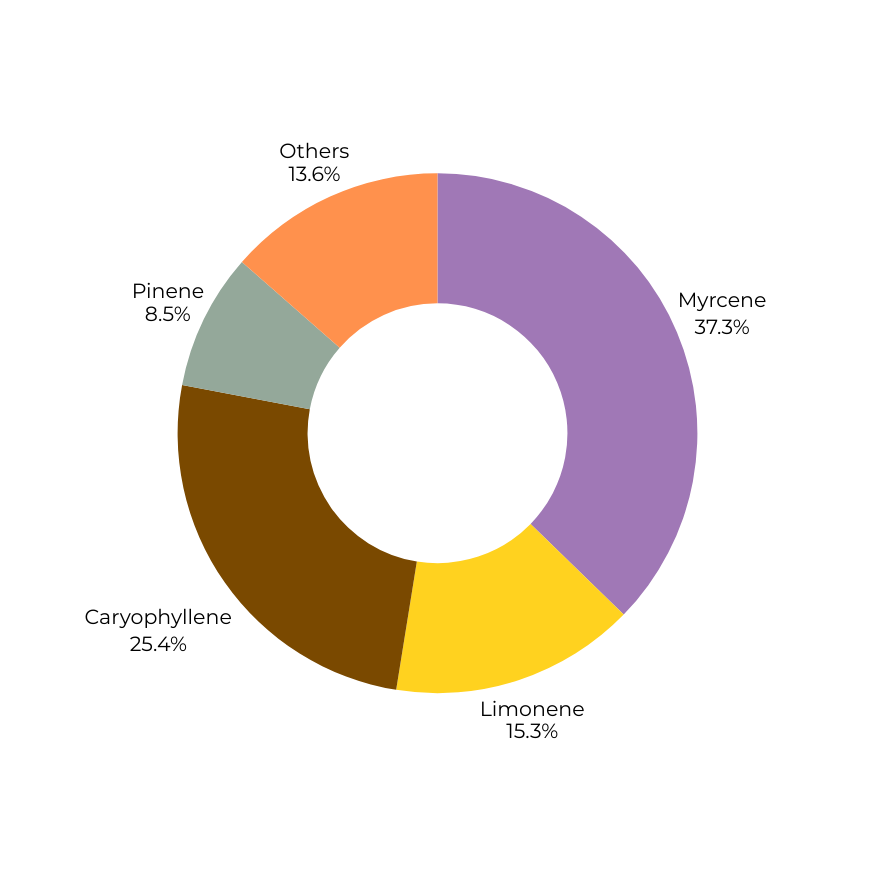
Flower A
+
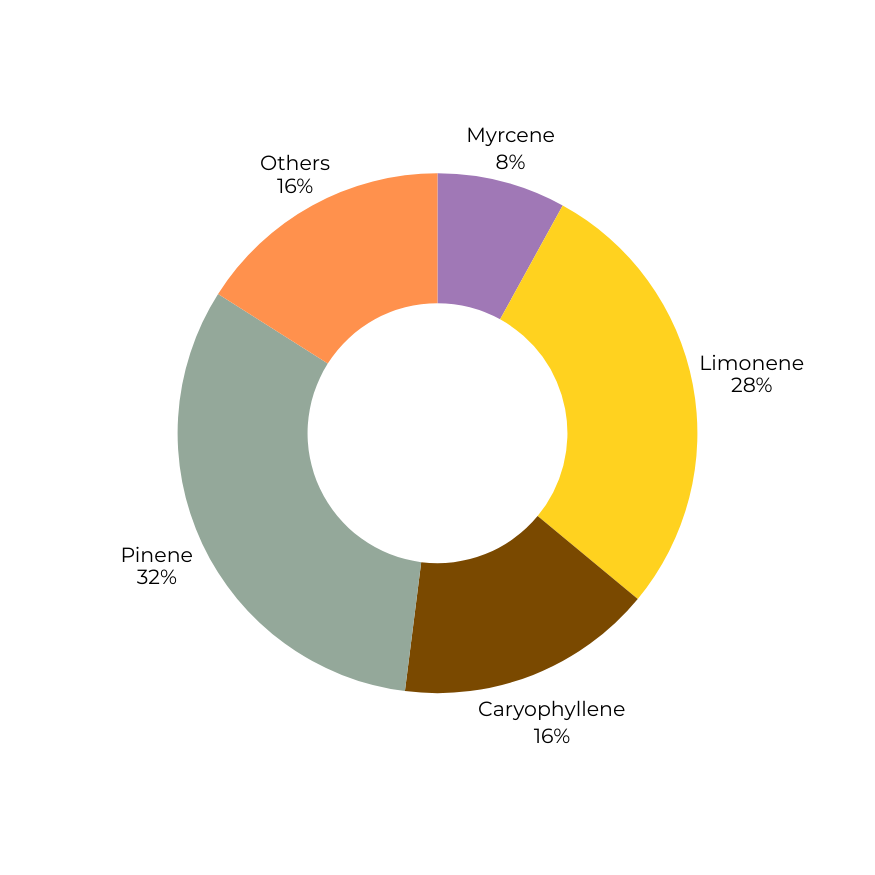
Flower B
Others
0.40%
0.4%
0.4%
Constructed Composite (1:1 by mass)
͢
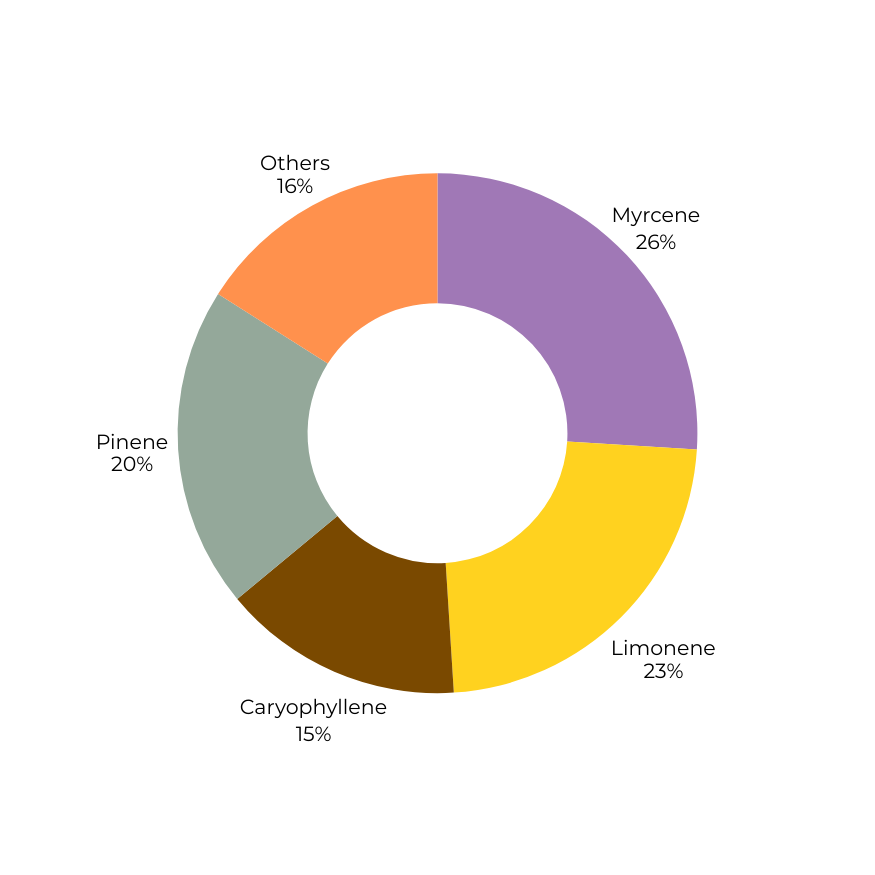
-
High myrcene weighting with secondary limonene support.
Tends toward heavier somatic tone, faster perceptual settling, and greater sedative bias at higher doses.
Dominance structure: singular and steep.
-
Elevated pinene and limonene with reduced myrcene presence.
Associated with brighter cognitive tone, sharper onset character, and greater perceptual clarity.
Dominance structure: alert-leaning and top-weighted.
-
Redistributed volatile weighting with reduced myrcene dominance and elevated pinene contribution.
Produces a more balanced directional profile, moderated somatic heaviness, and reduced extremity of onset bias.
Dominance structure: flattened and distributed.
Terpene
B
Composite
Myrcene
1.10%
0.2%
0.65%
Limonene
0.45%
0.70%
0.575%
Caryophy.
0.35%
0.4%
0.375%
Pinene
0.20%
0.8%
0.5%
Formula (for 1:1 blend by mass)
Composite % = (Flower A % + Flower B %) ÷ 2
For uneven blends (by mass):
Composite % = (A × mass₁ + B × mass₂) ÷ (mass₁ + mass₂)
Mathematical:
Composite % = Σ (component % × component mass) ÷ total mass
Volatile composition – as well as cannabinoid ratios – becomes a controllable variable.

Applied Cannabis Requires Structure.
Cannabis chemistry is measurable.
Compound release is controllable.
State response is modelable.
When these variables are structured together, psychoactivity becomes predictable.
Delta-Nine is not a product.
It is a methodology.
FAQs
-
No. Our systems complement medical consultation. They provide structure and clarity but should always be used alongside professional medical guidance.
-
Those categories are cultural shorthand, not science. Delta-Nine uses chemistry, pharmacology, and psychology to predict outcomes with precision, beyond vague labels.
-
Predictability doesn’t mean eliminating all variability — but it means replacing guesswork with structured rules. Our methods align compound behaviour with reproducible therapeutic effects.
-
Our system aligns with clinical evidence for pain, anxiety, spasticity, sleep, appetite regulation, and more — while also supporting wellness applications like focus and creativity.
-
A precision vaporiser (with temperature control) is recommended to apply the Tiered Vaporisation Protocol. However, we provide adaptations for common devices.
-
Yes. By structuring use and tying it to therapeutic intent, the framework reduces wasted use and supports healthier, more intentional patterns. For more serious concerns, we also offer guidance and referrals.
